Implantable foot device to restore feeling and reduce pain associated with diabetic nerve complications.
Problem:
Sixty to seventy percent of the 400 million diabetes patients worldwide suffer from nerve complications like distal sensory polyneuropathy (DSP), which is characterized by foot pain and loss of feeling. If left unchecked DSP progresses, resulting in gait instability, falls, and can even lead to amputation. Current treatments are limited, consisting of either pain medication or biofeedback devices to alleviate sensory loss. No individual therapeutic addresses both pain and sensory issues simultaneously. To further complicate matters, all current methods suffer from lack of patient compliance (as low as 36%).
Solution:
In response, the inventors developed a nerve stimulation device that treats both pain and loss of feeling. In addition, as an implantable device there are no compliance issues.
Technology Overview:
The inventors designed a minimally invasive implantable system to activate nerves in the leg in response to pressure on of the foot. The device is composed of two implantable components: a pressure sensor implanted under the skin on the bottom of the foot, and a battery-powered base unit implanted in the thigh connected to the leg nerves. When the foot sensors detect pressure, they wirelessly transmit signals to the base unit to provide nerve stimulation as therapy.
Advantages:
- Treats both the pain and loss of feeling associated with DPS simultaneously, unlike current treatments which target each concern separately
- Implant circumvents patient noncompliance, resulting in more comprehensive treatment
- Greatly reduces the risk of diabetic foot ulcers and diabetes-associated amputation
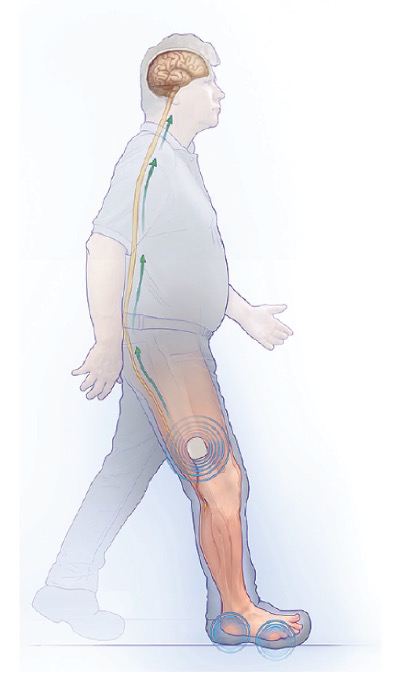
Proposed device for DSP. Pressure signals from subcutaneous sensors in the numb foot wirelessly drive electrical stimulation of peripheral nerves near the knee and result in therapeutic sensory feedback.
Stage of Development:
- Target Identified
- Preclinical Discovery
Case ID:
21-9530-tpNCS
Web Published:
3/4/2021
Patent Information:
| App Type |
Country |
Serial No. |
Patent No. |
File Date |
Issued Date |
Expire Date |