Co-expression of modified chimeric antigen receptor (CAR) T cells and cholesterol 25-hydroxylase (CH25H) in a single construct for treatment of solid tumor and hematological cancers.
Problem:
CAR T cell therapy is a treatment in which T cells collected from a patient are engineered to produce CARs that recognize and attack cancer cells. While it is efficacious at treating some patients with hematologic malignancies, many patients relapse and there is little success at treating solid tumors, which represent ~90% of adult cancers and 40% of childhood cancers. Cancer cells can evade CAR T therapy through trogocytosis, whereby an antigen on cancer cells is transferred to cytotoxic T lymphocytes (CTLs). This causes CTLs to become exhausted, be mistakenly recognized as targets and killed, and allows cancer cells to survive. Partial loss of CH25H in intratumoral T cells leads to increased trogocytosis and suppresses the viability and activity of native and therapeutic CAR T cells.
Solution:
The inventors designed constructs co-expressing CH25H with CARs that inhibit trogocytosis, prevent T cell exhaustion, and increase viability and tumoricidal activity of CAR T cells in both hematological and solid tumor cancers.
Technology:
Based on the discovery made by the inventors that downregulation of CH25H in CTLs diminishes CTL cytotoxicity and promotes trogocytosis, they engineered CARs to bicistronically express CH25H controlled by the EF1 alpha promoter within the CAR T constructs. Upon administration of the engineered CAR T cells to solid tumor and hematological cancer cells, the inventors observed decreased trogocytosis and a lesser extent of T cell exhaustion along with increased efficacy of antitumor effects. These findings highlight the ability of CH25H-co-expressing CAR T cells to bolster characteristics of CTLs that aid in their tumor killing and provide justification for the utilization of a CH25H CAR T approach to improve cancer immunotherapy.
Advantages:
- Decrease in solid tumor mass and volume, and increase of T cells in tumor and blood
- Greater killing activity, decrease in antigen transfer to CTLs, decrease in T cell exhaustion, and improved T cell viability and tumor infiltration
- Better persistence of CH25H-expressing CAR T cells in host, decreased number of leukemic cells in blood and prolonged animal survival
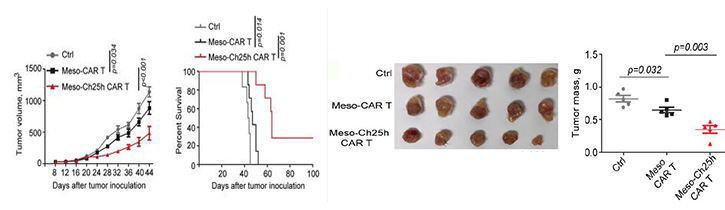
CARs designed to re-express CH25H inhibit trogocytosis and increase therapeutic efficacy.
Stage of Development:
- In vitro and in vivo proof of concept
Case ID:
22-9930-tpNCS
Web Published:
2/7/2023
Patent Information:
| App Type |
Country |
Serial No. |
Patent No. |
File Date |
Issued Date |
Expire Date |