One-pot combinatorial chemistry for fast and facile synthesis of amidine-incorporated degradable cationic lipids for lipid nanoparticle formulation and non-liver nucleic acid delivery.
Problem:
Messenger RNA (mRNA)-based therapeutics and vaccines have broad applications for a variety of diseases but require safe and efficient delivery mechanisms. Lipid nanoparticles (LNPs) are widely used for mRNA delivery and require an essential cationic lipid component for protecting and transporting the nucleic acid cargo. Current methods of synthesizing cationic lipids require complex multi-step reactions, long incubations, or extreme temperatures. Synthesis of cationic lipids with quaternary or tertiary amines are especially laborious, limiting the discovery of novel lipid candidates and the ability to scale up LNP manufacturing. Moreover, traditional cationic lipid-formulated LNPs mainly target the liver, necessitating the development of novel LNPs to enable the delivery of non-liver mRNA therapeutics.
Solution:
A combinatorial chemistry for amidine-incorporated degradable (AID) lipids presents an opportunity for rapid, high-yield cationic lipid synthesis. This approach can be conducted at room temperature in a one-pot reaction to generate over 100 amidine-incorporated degradable (AID) lipid. AID-lipids have not previously been well-studied but have high non-liver mRNA delivery capabilities as components of LNPs.
Technology:
A one-pot tandem reaction enables rapid synthesis of AID-lipids at room temperature. This reaction results in approximately 100 chemically diverse AID cationic lipids within an hour. Most AID-lipid LNPs (98%) effectively deliver mRNA in vitro and many act in vivo. Some AID-lipid LNPs produce more potent, systemic mRNA delivery than gold standard cationic lipids such as 1,2-dioleoyl-3-trimethylammonium-propane (DOTAP) and can transfect the liver, spleen, and lung. This synthetic methodology can be modified to generate lipids with different head and tail chemistries that can be screened for cargo delivery efficiencies.
Advantages:
- AID-lipids are synthesized in one hour at room temperature.
- Combinatorial chemistry for AID-lipids is conducted in a one-pot reaction.
- One reaction can synthesize >100 AID-lipids.
- AID-lipids outperform current gold-standard cationic lipids as components of LNPs and reach the lung, liver, and spleen.
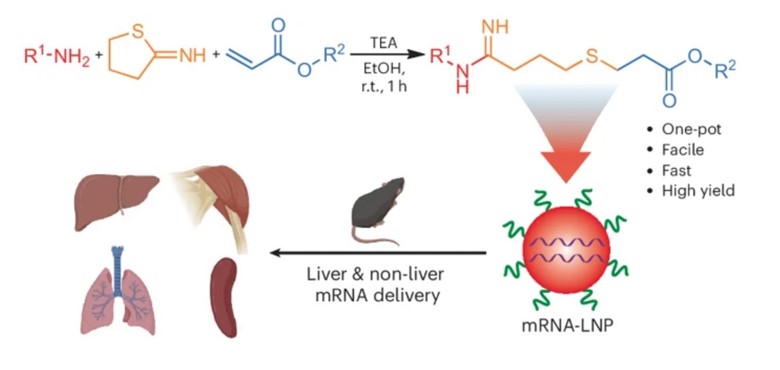
Illustration of the amine-thiol-acrylate conjugation reaction at room temperature (r.t.) for one hour in the presence of the base triethylamine (TEA) and ethanol (EtOH). Resulting lipids are formulated into mRNA-LNPs and their in vitro and in vivo mRNA delivery capabilities were tested.
Case ID:
23-10146-TpNCS
Web Published:
2/10/2026
Patent Information:
| App Type |
Country |
Serial No. |
Patent No. |
File Date |
Issued Date |
Expire Date |