Targeting Heat Shock Factor 1 (HSF1) via gene therapy or using pharmacological compounds targeting HSF1 (e.g. geranylgeranylacetone or macrocyclic peptides) can increase sleep stability, promote deeper sleep, reduce sleep fragmentation, and improve recovery from sleep loss.
Problem:
Millions of people worldwide suffer from disrupted sleep, whether due to modern "24/7" lifestyles, stress, aging and menopause, or underlying health conditions. Yet current treatments largely sedate patients without actually improving the quality of sleep - they can disrupt normal sleep architecture and fail to promote the deep, restorative stages that the brain and body need. This matters because poor sleep is not merely an inconvenience but is closely linked to cognitive decline, neurodegenerative disease, and reduced productivity. There is thus a critical need for therapies that restore how sleep naturally works, rather than simply masking symptoms.
Solution:
Dr. Reddy and his team showed that HSF1 overexpression in mice enhanced exhibited longer, uninterrupted sleep bouts and fewer awakenings. When mice were exposed to sleep deprivation, the mice overexpressing HSF1 exhibited enhanced recovery sleep efficiency which implies that HSF1 activation could mitigate the negative consequences of sleep loss, a feature with potential translational applications for individuals experiencing sleep deprivation.
Technology:
GGA: Geranylgeranylacetone (GGA) is a drug that has been safely used in Japan since 1984 to treat stomach ulcers. It has also been found to be a powerful activator of HSF1. With decades of clinical use and no reported neurological side effects, GGA is a strong candidate for repurposing to activate HSF1 in humans to improve sleep and treat sleep disorders. In mouse studies, GGA produced the same sleep benefits seen with direct HSF1 overexpression, including more stable sleep patterns and improved recovery after sleep loss. GGA shows the same HSF1-driven effects in fruit fly and roundworm sleep models, confirming that this mechanism is deeply conserved across species.
Macrocyclic peptides: Beyond GGA, the team is developing AI-designed macrocyclic peptides, ring-shaped molecules engineered to activate HSF1 with greater potency and precision. These macrocycles are Pareto-optimized, meaning multiple competing properties such as potency, selectivity, and safety are balanced simultaneously rather than trading one off against another. Using proprietary techniques, the designs target optimal ADME (absorption, distribution, metabolism, and excretion) and brain penetration in vivo.
Genetic expression: For severe sleep disorders linked to Alzheimer's, Parkinson's, or traumatic brain injury, AAV-mediated HSF1 overexpression could provide a one-time genetic intervention, with mRNA delivery as an adaptable alternative.
Together, these HSF1 activators open a broad market opportunity, from everyday sleep improvement for professionals, parents, and aging populations (e.g. menopausal women) to cognitive enhancement for executives, athletes, and students seeking maximum restoration from limited sleep.
Advantages:
- First-in-class mechanism
- Restoration of natural sleep architecture at the molecular level
- Mechanism conserved across species (mice, fruit flies, roundworms), increasing translational confidence
- Minimal side effects: HSF1 activation does not impair cognition, motor function, or induce next-day drowsiness
- Non-addictive mechanism of action, unlike benzodiazepines and related sedatives
- Does not suppress REM or other essential sleep stages, unlike many current sleep drugs
- Potentially accelerated path to market by utilizing clinically tested GGA
- GGA’s well-established safety profile and no reported neurological side effects
- Compatible with existing therapies for full sleep restoration
- Increase in sleep stability and reduction of sleep fragmentation
- Support of deeper sleep and improved recovery from sleep loss
- Potential to reduce total sleep time needed by improving restorative efficiency
- Broad therapeutic reach: applicable across lifestyle, wellness, and neurodegenerative disease markets
- Platform extensibility: GGA insights inform next-generation macrocyclic peptides and gene therapy approaches
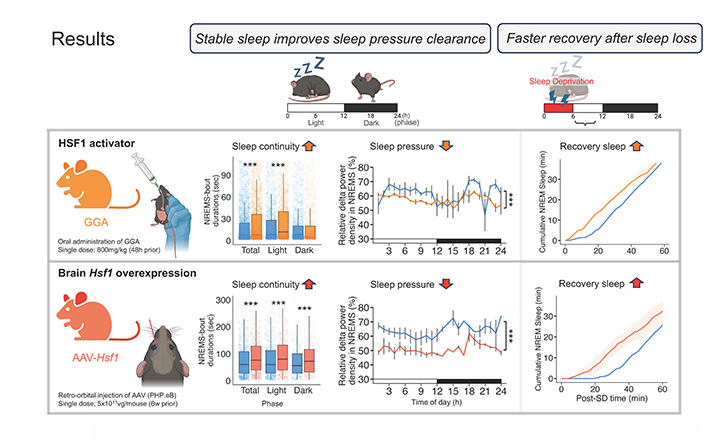
Activating the HSF1 pathway improves sleep quality and helps the brain bounce back faster after sleep loss.
Top row: a single oral dose of GGA, administered at a dose equivalent to levels already used safely in humans, suggesting there may be room to increase dosing for stronger effects.
Bottom row: a gene therapy delivering Hsf1 gene directly to the brain. First, sleep became more consolidated - mice slept in longer, uninterrupted bouts rather than waking frequently (left panels). Second, sleep pressure - the brain's built-up need for sleep, measured by slow-wave brain activity - dropped significantly, suggesting that sleep was more restorative (centre panels). Third, after being kept awake for several hours, treated mice fell back asleep faster and recovered more sleep in less time than untreated controls (right panels). ***p < 0.001. Blue, control; orange, GGA; red, Hsf1 gene therapy.