A flexible toolkit for human gene editing, activation, and repression that can be applied in orthogonally and in various disease models.
Problem:
There is increasing demand for technologies that can simultaneously edit the genome and regulate the transcriptome without relying on toxic double-stranded breaks. Currently, treating complex diseases such as cancer require correcting genomic point mutations while simultaneously activating or repressing secondary genes that are deregulated. These independent tasks require the co-delivery of multiple large effectors, drastically exceeding the strict packaging limits of modern clinical vectors like AAVs. Therefore, there is a dire need for a single, compact tool that provides orthogonal control over gene editing and expression. Such a unified platform would bypass viral packaging bottlenecks, eliminate CRISPR/Cas9-induced cytotoxicity, and unlock next-generation precision therapies.
Solution:
This invention is a minimal versatile genetic perturbation technology (mvGPT), which combines a prime editor (PE), fusion activator (MPH), and a multiplex array that produces RNA tailored for a variety of genetic perturbations, including genomic editing, gene activation, and gene repression. Importantly, unlike other currently available tools, mvGPT can be used orthogonally – such that gene activation, repression, and editing can be deployed independently and without interference among functions.
Technology:
The invention consists of three main components: an engineered compact prime editor (Prime Editor with Advanced Kernel, or PEAK), a transcriptional activator (MS2–p65–HSF1, or MPH), and an RNA system that produces short RNAs to direct editing, activation, or silencing of specific genes (drive-and-process array, or DAP). DAP, in combination with prime editing guide RNA (pegRNA) and nicking guide RNA (ngRNA), efficiently guides PEAK to target loci to modify DNA. MPH and PEAK are guided by short guide RNA to activate gene transcription. Finally, DAP encodes short hairpin RNA and facilitates gene repression through RNA interference.
Advantages:
- Orthogonal and independent deployment of gene editing, activation, and repression without interference
- Versatile delivery approaches of the mvGPT components (AAV, LV, mRNA, plasmids)
- Compatible with human cells and human disease models
- Does not result in DSB of DNA or cytotoxicity associated with existing gene editing methods
- Proof-of-concept model results in simultaneous 5% correction for the disease-causing gene ATP7B, a 1700-fold activation of the PDX1 gene, and a 93% repression of the TTR gene in a human cell line
- Can be utilized for any application requiring gene editing, activation, and/or repression
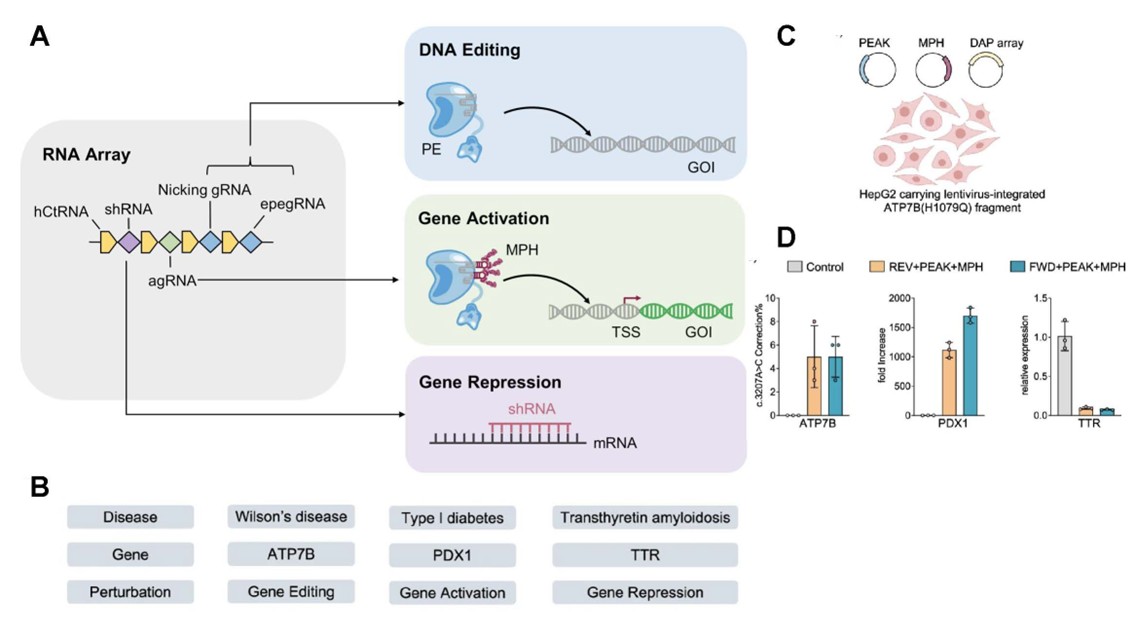
Simultaneous editing, activation, and silencing of disease-relevant genes using mvGPT in human liver cells. (A) Structure of the DAP RNA array encoding for nicking guide RNA (ngRNA) and engineered prime editing guide RNA (epegRNA) for gene editing, a truncated activation single guide RNA (agRNA) for gene activation, and a short hairpin RNA (shRNA) for gene silencing. (B) Schematic of a genetic disease model requiring orthogonal editing, activation, and silencing of a set of genes. This example exhibits a model involving Wilson’s disease, Type I diabetes, and Transthyretin amyloidosis. Rows indicate the disease, gene of interest, and the perturbation involved. (C) Illustration of HepG2 cells line (human liver cancer cells) transfected by plasmids encoding the three main elements of the mvGPT: prime editing system with advanced kernel (PEAK), fusion activator MS2–p65–HSF1 (MPH), and the drive-and-process (DAP) multiplexed RNA expression array. (D) mvGPT successfully corrected the ATP7B mutation in 5% of treated cells, increased PDX1 gene expression up to 1,700-fold, and reduced TTR gene expression by 93% -- all at once. FWD and REV indicate two versions of the DAP array with RNA components encoded in opposite order, both yielding comparable results.